Continuing the dialogue on women’s health

The first Cambridge Conversations of 2026 brought together leading researchers to explore the rapidly evolving field of women’s and maternal health.
Chaired by Professor Amanda Sferruzzi‑Perri, the discussion featured contributions from Dr Tereza Cindrova‑Davies, Dr Priscilla Day‑Walsh, and Dr Roser Vento‑Tormo, who shared insights into the remarkable complexity of the uterine environment and the biological, environmental and societal factors that shape maternal health across the life course.
The panel highlighted longstanding gaps in understanding conditions such as endometriosis and fibroids, alongside the urgent need for greater awareness, investment and innovation in women’s health.
Emerging technologies and interdisciplinary approaches, the speakers agreed, are now transforming research in women’s health, offering new opportunities to place women’s biology at the centre of health and wellbeing where it belongs.
The original discussion sparked wide interest, prompting us to bring the panel back together to revisit key themes and expand on the ideas raised. In this follow‑up, Professor Amanda Sferruzzi‑Perri shares the group’s collective reflections.
Complexity of the uterine environment
An active and delicately balanced system
The uterine environment is far more complex than it might first appear, and this complexity is central to the success or failure of early pregnancy. It is often surprising to learn that most human pregnancies end very early, frequently before a woman is even aware of conception.
Many of these losses occur at the point when the embryo attempts to implant into the lining of the uterus, known as the endometrium, or shortly thereafter. This reflects just how finely tuned the uterine environment must be.
Rather than acting as a passive surface, the endometrium is a dynamic and highly responsive tissue. It must be precisely prepared to support the embryo, engaging in a coordinated exchange that supports implantation and guides early development. This involves not only structural readiness but also active communication and nutritional support.
For example, specialised endometrial glands secrete key factors that nourish the embryo before the placenta is fully established, a process known as histotrophic nutrition. This early support plays a crucial role in laying the foundations of the placenta, which will later sustain the pregnancy.
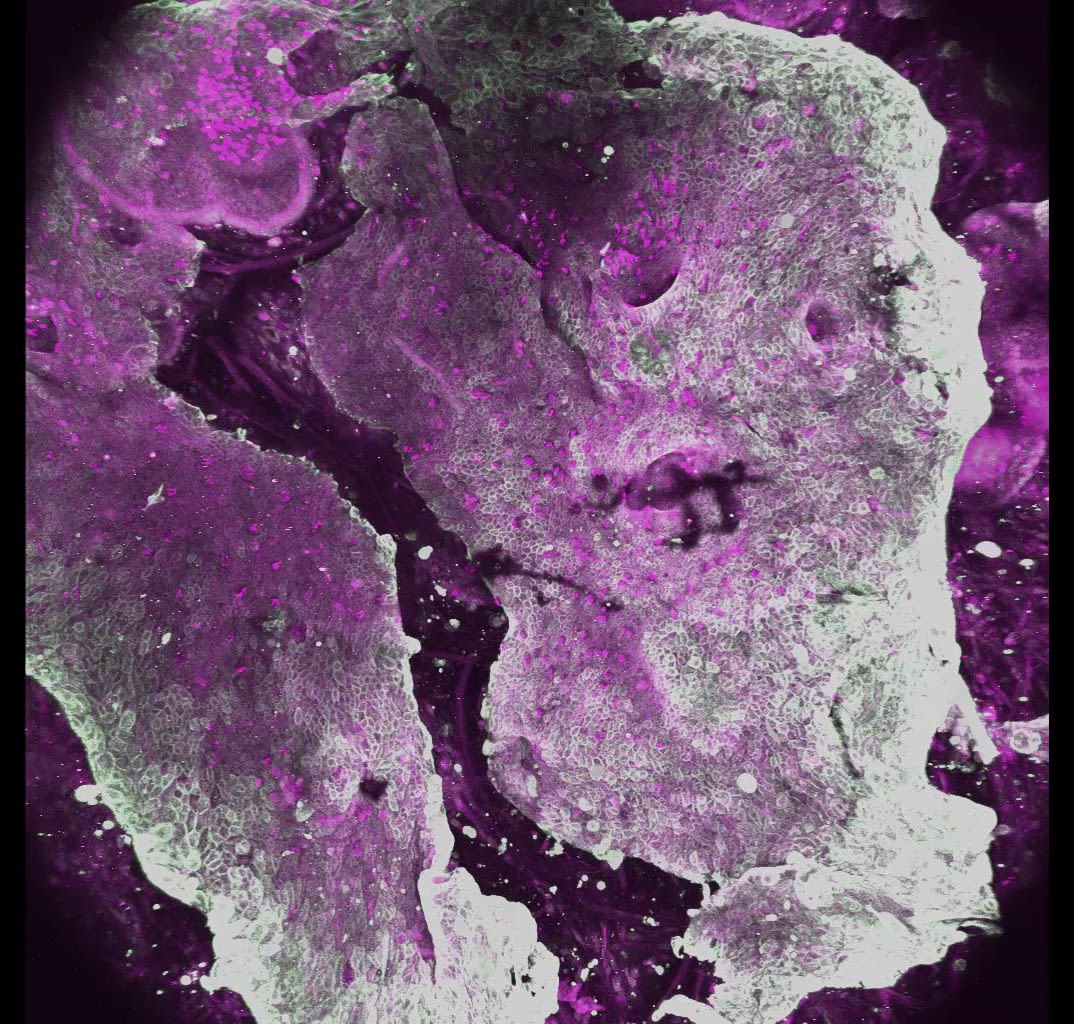
Model of the human endometrium, generated using endometrial organoids and stromal cells, image media provided by Dr Tereza Cindrova-Davis
What makes this environment particularly complex is that early events set the trajectory for the entire pregnancy. The first weeks following implantation represent a critical “blueprint” phase, during which placental cell types form and maternal–fetal interactions are established. Disruptions at this stage may remain silent for months, only becoming apparent later as complications such as miscarriage, failed implantation, or conditions like pre-eclampsia.
Increasing evidence suggests that, in some cases, these outcomes originate from subtle abnormalities in how the endometrium prepares for and responds to pregnancy, an idea sometimes described as an “endometrial spectrum” of disorders.
Adding another layer of complexity is the immune environment of the uterus. Pregnancy presents a unique biological challenge: the embryo carries genetic material from both parents and is therefore partly foreign to the mother’s immune system. Instead of mounting a typical defensive response, the uterus creates a highly specialised local immune setting. Certain immune cells that could be harmful are actively excluded (e.g. B and T cells), while others are adapted to support pregnancy (e.g. UNK cells).
These resident immune cells do not simply defend against infection; they take on regulatory and developmental roles. They help guide placental cells as they invade the maternal uterus and assist in reshaping uterine blood vessels to ensure an adequate supply of oxygen and nutrients to the growing fetus. This process depends on finely balanced signalling between maternal immune cells and foetal placental cells, with variations in these interactions influencing pregnancy outcomes.
Crucially, the uterine environment is not uniform. Its integrity matters greatly. When implantation occurs in areas where the endometrium is damaged or altered, such as over scar tissue, the risk of severe complications increases. This highlights the essential role of both the structural and immune components of the endometrium.
Taken together, these insights reveal that the uterine environment is a highly coordinated system involving hormonal, cellular, and immune elements working in concert. It is not merely a site of implantation, but an active, adaptive interface that supports, regulates, and can determine the course of pregnancy.
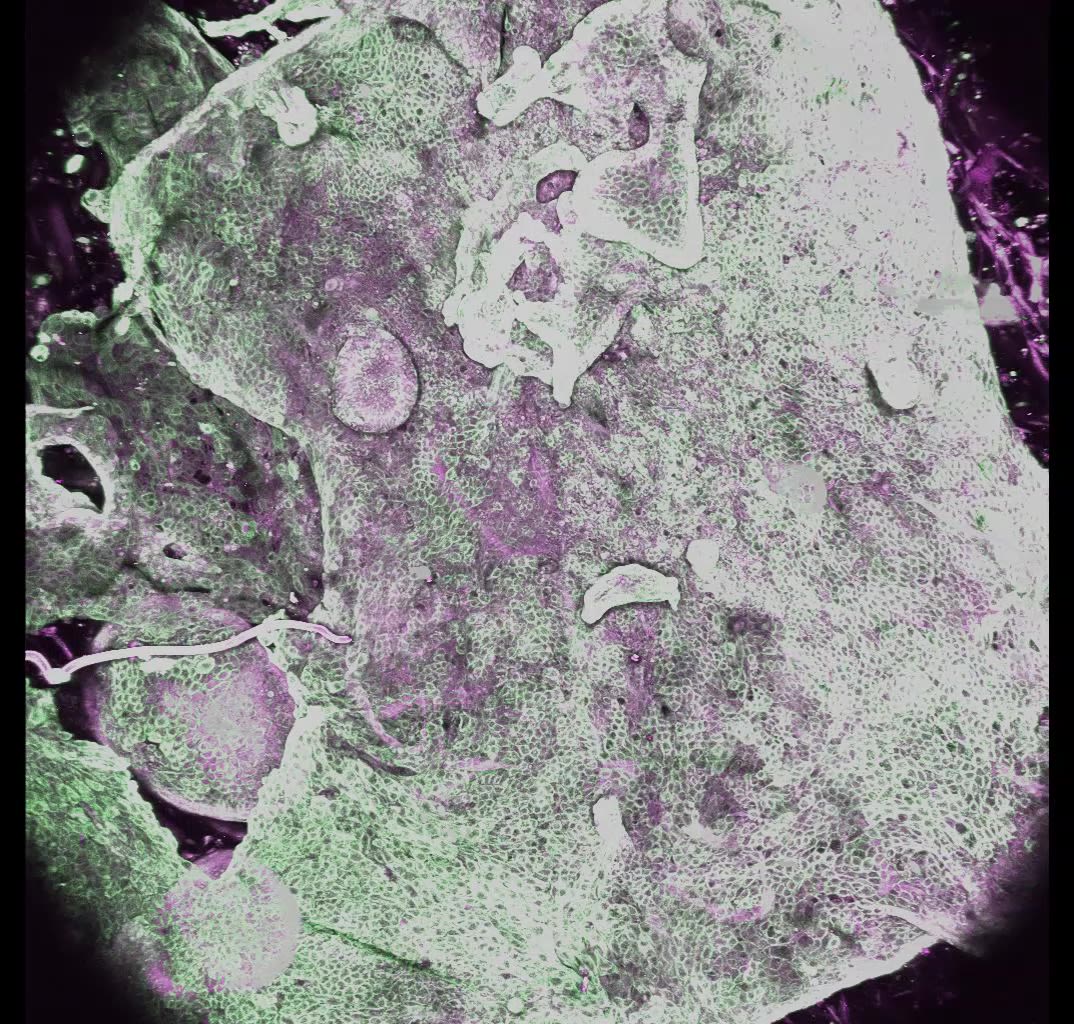
Human endometrial surface in vivo, image provided by Dr Tereza Cindrova-Davis
Human endometrial surface in vivo, image provided by Dr Tereza Cindrova-Davis
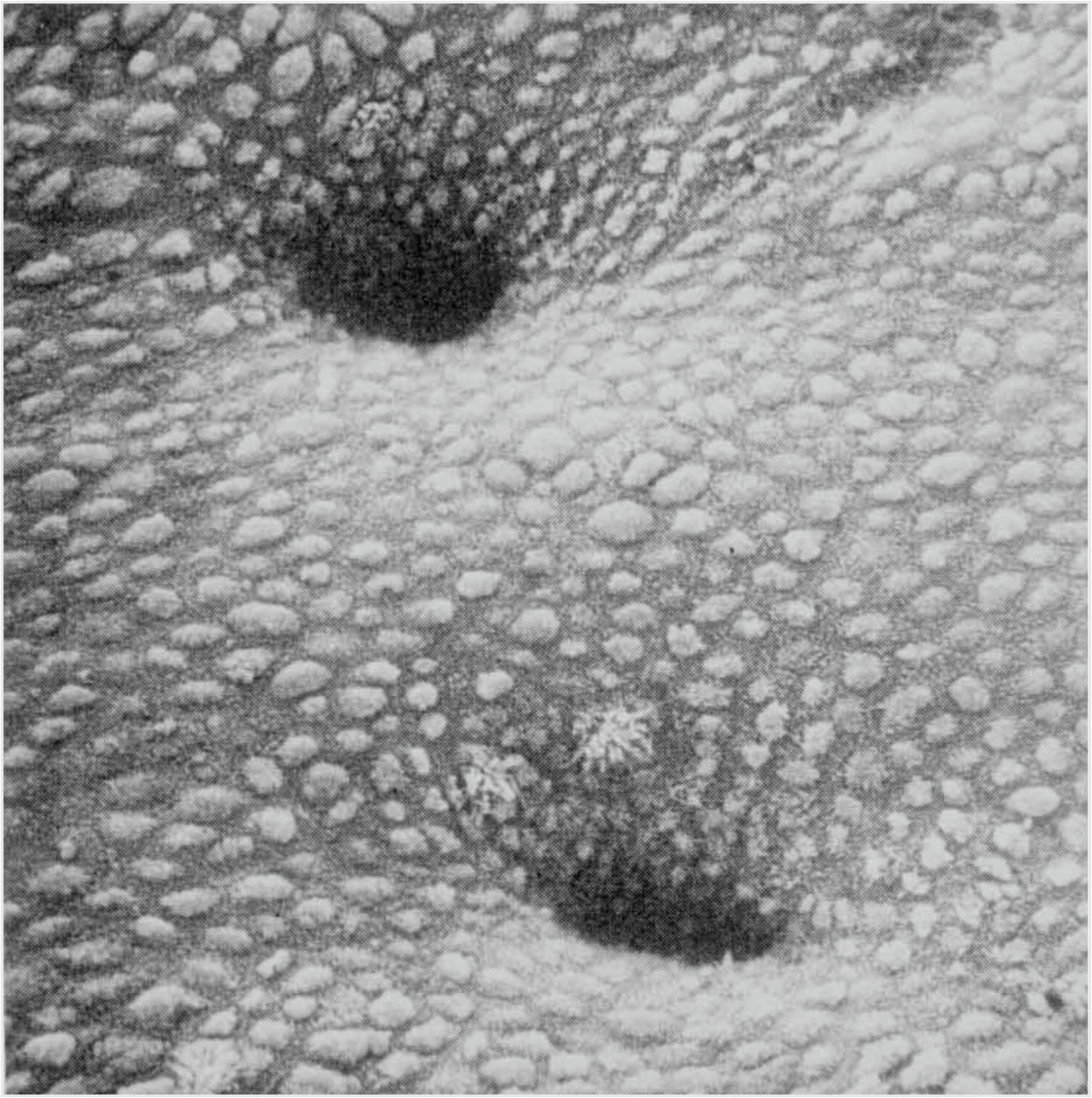
Endometrial lining - in vitro model image provided by Dr Tereza Cindrova-Davis
Endometrial lining - in vitro model image provided by Dr Tereza Cindrova-Davis
Biological, environmental, and societal influences on maternal health

Dr Priscilla Day-Walsh presenting at the All-Party Parliamentary Groups
Dr Priscilla Day-Walsh presenting at the All-Party Parliamentary Groups
Professor Amanda Sferruzzi‑Perri pictured with doctors, scientists, and healthcare workers in Kenya, where research is focused on understanding the high prevalence of adverse maternal outcomes.
Professor Amanda Sferruzzi‑Perri pictured with doctors, scientists, and healthcare workers in Kenya, where research is focused on understanding the high prevalence of adverse maternal outcomes.
From biology to inequality
Maternal health is shaped by a complex interplay of biological, environmental, and societal influences, all of which interact across pregnancy and throughout a woman’s life.
Biologically, pregnancy is a whole-body process that requires careful coordination between multiple systems, including the cardiovascular, metabolic, endocrine, and immune systems. The maternal body undergoes significant adaptations to support the developing fetus, with changes in blood flow, shifts in insulin sensitivity, and finely tuned immune regulation.
These adaptations are partly orchestrated by the placenta, a temporary organ that acts as the communication interface between mother and fetus, secreting signals, including hormones and inflammatory mediators, that reshape maternal physiology.
While many of these adjustments resolve after birth, they are not always fully reversible. In some cases, pregnancy can leave lasting effects on organs such as the heart, blood vessels, and pancreas, influencing a woman’s long-term health. Complications such as pre-eclampsia and gestational diabetes reflect disruptions in these adaptations and are increasingly recognised as early indicators of future cardiovascular and metabolic disease.
Environmental factors further shape these biological processes. Modifiable risks such as over-nutrition, obesity, and substance use increase the likelihood of pregnancy complications, particularly hypertensive disorders.
In addition, broader exposures, including chronic stress, infection, and extreme heat, have been linked to outcomes such as preterm birth, stillbirth, and postpartum depression. These influences can act both directly, by affecting maternal physiology, and indirectly, by altering how the body responds to pregnancy.
Crucially, these biological and environmental risks are embedded within a wider societal context. Maternal health outcomes are not evenly distributed, and disparities persist across different populations.
For instance, Black women are approximately twice as likely to develop pre-eclampsia compared to white women. They also experience higher rates of severe complications, including kidney and heart problems, and an increased risk of death. Higher rates of underlying conditions such as hypertension, diabetes, and anaemia contribute to this risk, but they do not fully explain the differences observed.
Inequities in access to and quality of healthcare, along with socioeconomic disadvantage, can delay the diagnosis and management of complications. At the same time, chronic stress, particularly that associated with structural inequalities and discrimination, has measurable physiological effects that can negatively influence pregnancy outcomes.
There is also evidence that genetic and immunological variation may contribute to differences in risk between populations, interacting with environmental and social factors in complex ways. This underscores the importance of studying diverse groups and recognising that maternal health is shaped by multiple, overlapping influences rather than a single cause.
Despite growing awareness, women’s health research remains significantly underfunded; in 2020, only about 5% of global R&D funding was allocated to this area, and just 1% of that supported non-cancer conditions.
Taken together, these insights highlight that maternal health cannot be understood through biology alone. It reflects a dynamic interaction between the body, the environment, and the social conditions in which women live. Improving outcomes, therefore, requires a more integrated approach; one that combines biological research with attention to environmental exposures and a commitment to addressing health inequalities across the lifespan.
Need for greater awareness and innovation

How urgent is the need for greater awareness and innovation around conditions such as endometriosis and fibroids?
There is an urgent need for greater awareness, earlier diagnosis, and innovation in the management of chronic gynaecological conditions such as endometriosis and uterine fibroids, both of which affect millions of women worldwide yet remain under-recognised and under-researched.
Endometriosis affects around 10% of women of reproductive age, characterised by tissue similar to the lining of the uterus growing outside the womb, often on the ovaries, bowel, or bladder.
Despite its prevalence and the profound impact it can have on pain, fertility, and quality of life, diagnosis is often significantly delayed, with women waiting an average of eight years for answers. This delay reflects both the biological complexity of the condition and a broader lack of awareness among healthcare systems and society more widely.
The causes of endometriosis are still not fully understood, which highlights the pressing need for further research. Current evidence suggests that both genetic and biological factors contribute, including pathways linked to inflammation, immunity, hormonal activity, metabolism, tissue stiffness, and even the microbiome. Several theories exist to explain how endometrial-like tissue develops outside the uterus, ranging from retrograde menstruation to the transformation of local stem cells within the pelvic cavity.
Researchers are now developing advanced laboratory and animal models, alongside studying patient-derived biological samples, to better understand disease mechanisms and identify biomarkers in blood, urine, or menstrual fluid that could enable earlier, less invasive diagnosis.
Alongside academic research, innovation is also emerging in women’s health technology. Several femtech companies founded by Cambridge scientists are working to improve the diagnosis and management of conditions such as endometriosis, infertility, and PCOS. These innovations, including AI-driven diagnostic tools and menstrual blood-based approaches, have the potential to dramatically reduce diagnostic delays and improve patient outcomes.
A similarly urgent issue surrounds uterine fibroids, which are non-cancerous growths of the uterus that affect up to two-thirds of women, with some studies suggesting that 70–80% of women will develop them by the age of 50. The burden is even greater among Black women, where prevalence may reach 80–90%, and symptoms are often more severe.
Fibroids can cause heavy menstrual bleeding, severe pain, infertility, iron-deficiency anaemia, pelvic pressure, urinary symptoms, and complications during pregnancy. These symptoms can lead not only to serious physical morbidity but also to social isolation, loss of income, and significant psychological distress.
Despite this, fibroids are frequently diagnosed late, often only when symptoms have become severe enough to require invasive assessment and treatment. Too often, symptoms such as heavy bleeding or pelvic pain are minimised because fibroids are described as “benign,” delaying intervention and increasing the likelihood of hysterectomy.
For many women, particularly Black women and those from marginalised communities, diagnostic delays can stretch over years due to slow referral pathways, inadequate menstrual health education, and systemic bias in clinical care.
Innovation in fibroid care is urgently needed, particularly in the development of fertility-preserving treatments, clearer decision-support tools, and standardised clinical training. At present, treatment pathways are often inconsistent, and access to uterus-preserving options remains unequal. The lack of dedicated national funding for fibroid research further compounds these challenges, leaving major gaps in both evidence and service provision.
The FibroidsFree, Cambridge team
The FibroidsFree, Cambridge team
Taken together, endometriosis and fibroids highlight a broader issue in women’s health: common, life-altering conditions are too often normalised, dismissed, or diagnosed too late.
Greater awareness must be matched by investment in research, innovation, and equitable care pathways. Improving outcomes for women will require not only scientific advances, but also a cultural shift in how menstrual pain, heavy bleeding, and reproductive symptoms are recognised and taken seriously.
Emerging technologies transforming research in women’s health
Technological breakthroughs in early pregnancy research
Emerging technologies are rapidly transforming research in women’s health, particularly in areas that have historically been difficult to study, such as the early stages of pregnancy and the uterine environment.
Much of what happens during this critical window occurs deep within the body and has, until recently, been largely inaccessible.
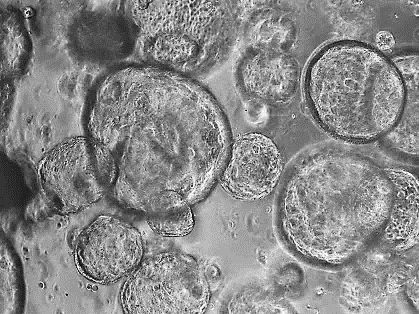
One major advance has been the development of organoid models, three-dimensional structures grown from patient cells that closely resemble real human tissues. In reproductive research, scientists can now generate miniature versions of endometrial glands in the laboratory. These models respond to hormones and behave in ways that closely reflect natural tissue, providing a powerful tool to study how the uterus prepares for pregnancy.
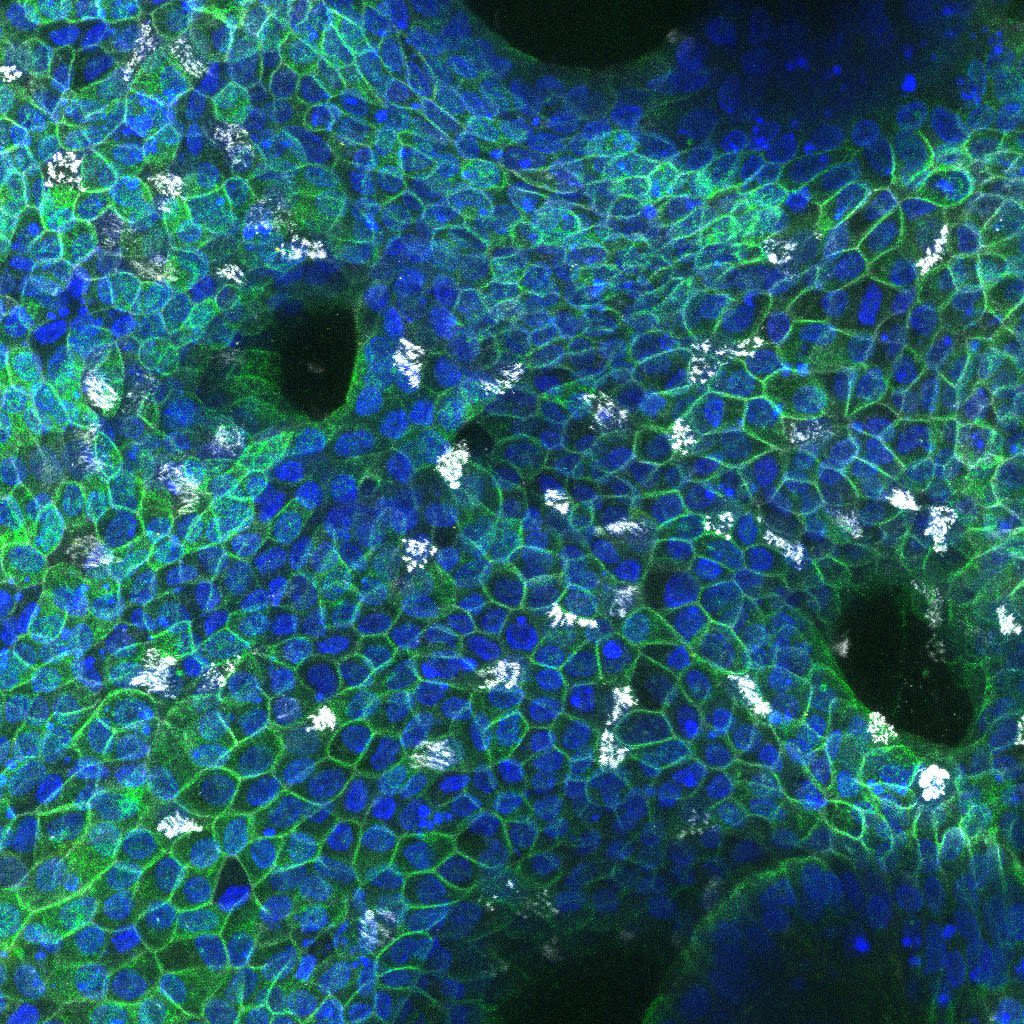
Human mini-organs - organoids - growing in culture
Human mini-organs - organoids - growing in culture
A key breakthrough has been the ability to create these organoids using cells collected non-invasively from menstrual fluid, rather than through biopsies. This approach makes it easier to study uterine biology across a wider range of individuals and opens the door to more personalised research into conditions such as implantation failure, miscarriage, and endometriosis.
These models also allow researchers to study the earliest stages of pregnancy, when many complications first begin. By recreating aspects of the uterine environment in the lab and growing different cell types together, scientists can investigate how maternal tissues, immune cells, and early placental cells interact. This provides new insight into the cellular “conversation” that underpins successful pregnancy.
At the same time, advances in genomic and computational technologies are providing unprecedented resolution. Techniques such as single-cell and spatial genomics allow researchers to analyse individual cells in detail, identifying their type, activity, and origin, as well as mapping their spatial coordinates and how they communicate with one another. This is helping to reveal the complex signalling networks that regulate implantation and early development.
High-risk longitudinal studies, such as the Pregnancy Outcome Prediction Studies (POPs 1 and POPs 2) in Cambridge, are also essential in characterising molecular markers and therapeutic targets for predicting, preventing and treating pregnancy complications.
Together, these technologies are enabling earlier, more precise, and less invasive investigation of women’s health. By allowing researchers to study processes that were previously out of reach, they are helping to uncover the underlying causes of reproductive disorders and pregnancy complications, and paving the way for improved diagnosis, prevention, and treatment.
Paradigm shift

What has been the most paradigm-shifting research in women’s health so far this century?
If this century’s advances in women’s health research could be distilled into a single paradigm shift, it would be the recognition that women’s biology is not a niche area of medicine - it is fundamental to understanding human health.
Over the past two decades, research has fundamentally challenged long-standing assumptions. It has revealed the extent to which women were historically underrepresented in clinical trials, leading to gaps in drug development, diagnosis, and treatment. It has also shown how conditions such as cardiovascular disease were long mischaracterised as primarily “male,” despite being a leading cause of death in women globally.
At the same time, maternal mortality and persistent ethnic disparities in outcomes are now increasingly understood not as inevitable biological differences, but as the result of systemic inequities in healthcare, research, and society.
These insights have driven meaningful change. They have reshaped research policy, influenced clinical guidelines, and increased awareness of sex-specific differences in disease presentation and progression. Importantly, they have also begun to correct the long-standing imbalance in how evidence is generated and applied in medicine.
Alongside these structural changes, scientific breakthroughs have transformed how we understand women’s health across the life course.
Advances such as HPV vaccination have demonstrated the power of prevention at scale. Growing recognition of perinatal mental health has reframed how we think about psychological well-being during and after pregnancy. And perhaps most importantly, pregnancy itself is now increasingly understood as a “window” into long-term health, where complications can signal future risk of cardiovascular, metabolic, or mental health disease.
Taken together, these developments have led to a broader conceptual shift: pregnancy, menstruation, menopause, and reproductive health are no longer seen as isolated biological events, but as interconnected processes that shape lifelong health trajectories.
Perhaps the most transformative insight of all is this reframing of time; seeing reproductive health not as a series of short, separate episodes, but as a continuous biological narrative that influences health across decades. This has opened new opportunities for prevention, earlier intervention, and a more integrated approach to medicine.
"Women’s biology has historically been under‑researched, despite its fundamental importance to human health. Addressing that imbalance is not optional, it’s essential."
Despite this progress, the transformation is still underway. The next paradigm shift will be fully realised only when women’s health is no longer treated as a specialised or peripheral field, but as a core pillar of biomedical research, clinical practice, and health policy. Efforts in centres such as Cambridge are helping to accelerate this change by bringing together biology, clinical insight, and equity-focused research to reshape how women’s health is understood and delivered
Keep the conversation going
Continue the conversation about women's health
To learn more about how philanthropic support can advance vital research into women’s health at the University, please contact Holly Singlehurst, Associate Director Biological Sciences.
Cambridge Conversations: Maternal health webinar
The first Cambridge Conversations of 2026 brought together leading researchers to explore the rapidly evolving field of women’s and maternal health. Chaired by Professor Amanda Sferruzzi‑Perri, the discussion featured contributions from Dr Tereza Cindrova‑Davies, Dr Priscilla Day‑Walsh, and Dr Roser Vento‑Tormo
Breaking the Silence about Uterine Fibroids: A Pathway to Legislative Change
Uterine fibroids affect millions, yet they remain underdiagnosed, underfunded, and largely overlooked in public health priorities Building on last year's Big Conversation on inequalities in women's health, this symposium shifts the focus from awareness to action.

Further reading
Networks
- Cambridge Reproduction (interdisciplinary centre)
- Women’s Biology and Health (network)
- Endometrial network (network)
Crucial publications and studies
- Underrepresentation of females in scientific research has significant consequences for disease treatment
- Mechanisms of implantation: strategies for successful pregnancy. Cha J, Sun X, Dey SK (2012) Nature Medicine 18: 1754–1767
- Histotrophic nutrition and the placental-endometrial dialogue during human early pregnancy. Burton, GJ, Cindrova-Davies T, Turco MY (2020) Placenta 102: 21-26
- Human placental development and function. Cindrova-Davies T and Sferruzzi-Perri AN (2022) Seminars in Cell and Developmental Biology 131:66-77
- Mapping the temporal and spatial dynamics of the human endometrium in vivo and in vitro. Garcia-Alonso L et al., (2021) Nature Genetics 53:1698-1711
- The Impact of Preeclampsia and Gestational Diabetes on Future Maternal Cardiometabolic Health. Barrell AM and Sferruzzi-Perri AN (2025). Acta Physiologica 241:e70113
- KIR2DS5 allotypes that recognise the C2 epitope of HLA-C are common among Africans and absent from Europeans. Blokhuis JH et al., (2017) Immun Inflamm Dis. 5:461-468
- Pregnancy, parturition and preeclampsia in women of African ancestry. Nakimuli A et al., (2014) Am J Obstet Gynecol. 210:510-520.e1
- www.zero.cam.ac.uk/node/616
- www.joh.cam.ac.uk/about-us/news-and-research/ps2m-boost-for-research-to-prevent-malaria-deaths-in-pregnancy
- Spatiotemporal cellular map of the developing human reproductive tract. Lorenzi V, et al., (2026) Nature 650:428-437
- The human endometrium: atlases, models, and prospects. Wong F, Kim CE, Garcia-Alonso L, Vento-Tormo R (2025) Curr Opin Genet Dev 92:102341
- Long-term, hormone-responsive organoid cultures of human endometrium in a chemically defined medium Turco MY et al., (2017) Nat Cell Biol. 19:568-577
- Menstrual flow as a non-invasive source of endometrial organoids. Cindrova-Davies et al., (2021) Comm Biol. 4:651
- Modelling human embryo implantation in vitro. Mole MA et al., (2026) Cell 189:87-105.e28
- Trophoblast organoids as a model for maternal–fetal interactions during human placentation. Turco TY et al., (2018) Nature 564:263-267
- Screening for fetal growth restriction with universal third-trimester ultrasonography in nulliparous women in the Pregnancy Outcome Prediction (POP) study: a prospective cohort study. Sovio U, White IR, Dacey A, Pasupathy D, Smith GCS. (2015) Lancet 386(10008): 2089-97
- Prediction of Preeclampsia Using the Soluble fms-Like Tyrosine Kinase 1 to Placental Growth Factor Ratio: A Prospective Cohort Study of Unselected Nulliparous Women. Sovio U, Gaccioli F, Cook E, Hund M, Charnock-Jones DS, Smith GC. (2017) Hypertension 69: 731-8
Policy-related documents
- committees.parliament.uk/committee/81/health-and-social-care-committee/news/209309/black-women-still-ignored-in-maternity-care-say-mps
- publications.parliament.uk/pa/cm5803/cmselect/cmwomeq/94/report
- MBRACE-UK. Saving lives, Improving mothers’ care: Lessons learned to inform maternity care from the UK and Ireland confidential enquiries into maternal deaths and morbidity 2018–20’. November 2022 (https://www.npeu.ox.ac.uk/mbrrace-uk/reports/maternal-reports/maternal-report-2018-2020)
- (ONS) OfNS. Births and infant mortality by ethnicity in England and Wales: 2007 to 2019. 2021 (https://www.ons.gov.uk/peoplepopulationandcommunity/healthandsocialcare/childhealth/articles/birthsandinfantmortalitybyethnicityinenglandandwales/2007to2019)
- ONS) OfNS. statistical bulletin, Child and infant mortality in England and Wales: 2023. 2024 (https://www.ons.gov.uk/peoplepopulationandcommunity/birthsdeathsandmarriages/deaths/bulletins/childhoodinfantandperinatalmortalityinenglandandwales/2023)
- www.homerton.cam.ac.uk/events/breaking-silence-about-uterine-fibroids-pathway-legislative-change
- publications.parliament.uk/pa/cm5803/cmselect/cmwomeq/1611/report
- Fibroids Report – CAHN & Black women with fibroids face delays and poor care in the UK, says report | Health | The Guardian